Fluorosilicic acid
Agent Name
Fluorosilicic acid
Alternative Name
Fluosilicic acid
CAS Number
16961-83-4
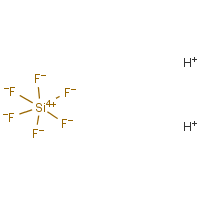
Formula
F6-Si.2H
Major Category
Toxic Gases & Vapors
Synonyms
Fluosilicic acid; Acide fluorosilicique [French]; Acide fluosilicique [French]; Acido fluosilicico [Italian]; Dihydrogen hexafluorosilicate; Dihydrogen hexafluorosilicate (2-); FKS; Hexafluorokieselsaeure [German]; Hexafluorokieselsaiure [German]; Hexafluorokiezelzuur [Dutch]; Hexafluorosilicic acid; Hexafluosilicic acid; Hydrofluorosilicic acid; Hydrofluosilicic acid; Hydrogen hexafluorosilicate; Hydrosilicofluoric acid; Kiezelfluorwaterstofzuur [Dutch]; Sand acid; Silicate (2-), hexafluoro-, dihydrogen; Silicofluoric acid; Silicofluoride; Silicon hexafluoride dihydride; Silicate(2-), hexafluoro-, hydrogen (1:2); [ChemIDplus] UN1778
Category
Acids, Inorganic
Description
Colorless liquid with a sour, pungent odor; [HSDB] Aqueous solution (<=35% fluorosilicic acid): Clear light yellow liquid; [Aldrich MSDS]
Sources/Uses
Used as a chemical intermediate, a disinfectant, a water fluoridating agent, a wood preservative, a masonry and ceramic hardener, and a glass additive; it is also used to treat hides and skins, to electroplate chromium, and to electrolytically refine lead; [HSDB] Used in technical paints, oil well acidizing, and to remove mold, rust, and stains from textiles; [NTP]
Comments
Corrosive to skin; [Quick CPC] High inhalation exposure may cause pulmonary edema; Potential for fluorosis after chronic ingestion or inhalation of aerosol; [ICSC] Marketed strictly as an aqueous solution; Anhydrous form dissociates almost instantly into silicon tetrafluoride and hydrofluoric acid; 60-70% solutions solidify at about 19 deg C, forming a crystalline dihydrate; [Merck Index] Fumes in air; Evolves heat and corrosive fumes on mixing with water; [CAMEO] Causes burns to eyes and skin (second-degree after contact of several minutes); Chronic exposure hazards include osteofluorosis, respiratory impairment, and injury to liver and kidneys; [CHRIS] Chronic exposure may cause changes in bone, corrosion of mucous membranes, coughing, shock, pulmonary edema, fluorosis, coma, and death; [NTP] Causes burns; Inhalation may cause corrosive injuries to upper respiratory tract and lungs; Toxic by ingestion; [Aldrich MSDS] See "FLUORIDES."
Biomedical References
Exposure Assessment
BEI
Fluorides in urine = 2 mg/L prior to shift or 3 mg/L at end of shift; (Repeated measurements recommended.)
TLV (ACGIH)
2.5 mg/m3, as F
PEL (OSHA)
2.5 mg/m3, as F
MAK
1 mg/m3, as F, inhalable fraction
IDLH (NIOSH)
250 mg/m3, as F
Reference Link #2
Adverse Effects
Toxic Pneumonitis
Yes
Dermatotoxin
Skin burns
Diseases, Processes, and Activities Linked to This Agent
Processes
Industrial Processes with risk of exposure: