Aluminum bromide
Agent Name
Aluminum bromide
CAS Number
7727-15-3
Formula
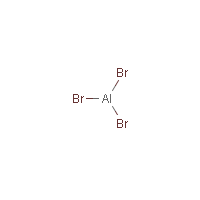
Al-Br3
Major Category
Metals
Synonyms
Aluminum bromide, anhydrous; Aluminum tribromide; Tribromoaluminum; [ChemIDplus] UN1725;
Category
Metals, Inorganic Compounds
Description
Whitish-yellow to yellowish-red crystals or lumps with a pungent odor; Deliquescent; Fumes in moist air; [CAMEO]
Sources/Uses
Used in organic synthesis; [HSDB]
Comments
Contact with substance may cause severe burns; Decomposes when burned to form corrosive gases; Reacts with water to form increased concentration of fumes; Experiments show that "there is essentially no HBr gas formation; the substance is quite unreactive." [CAMEO] Causes tissue burns; Oral LD50 (rats) = 1598 mg/kg; [HSDB] Corrosive; Harmful if swallowed; Reacts violently with water; Inhalation can cause chemical pneumonitis; [MSDSonline] See "Bromine." See "Aluminum."
Biomedical References
Exposure Assessment
TIH
Yes
Dangerous When Wet
Yes
Explanatory Notes
VP = 1 mm Hg at 81.3 degrees C; [HSDB] The Guide from the Emergency Response Guidebook is for "Aluminum bromide, anhydrous." Not TIH in land-based spills, but TiH substance released when spilled in water; [ERG 2016]
Adverse Effects
Toxic Pneumonitis
Yes
Dermatotoxin
Skin burns
Diseases, Processes, and Activities Linked to This Agent
Diseases
Occupational diseases associated with exposure to this agent:
Processes
Industrial Processes with risk of exposure: