Chromium(II) chloride
Agent Name
Chromium(II) chloride
CAS Number
10049-05-5
Formula
Cl2-Cr
Major Category
Metals
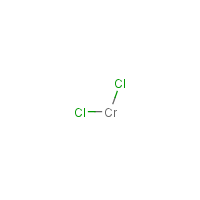
Synonyms
Chromium chloride (CrCl2); Chromium dichloride; Chromium(II) chloride (1:2); Chromous chloride; [ChemIDplus]
Category
Chromium Compounds, Inorganic
Description
Very hygroscopic solid; [Merck Index] Steel-gray lustrous crystals or powder; [MSDSonline]
Sources/Uses
Used in chromium plating and as a catalyst, oxygen absorbent, and textile mordant; [HSDB]
Comments
"The relatively unstable divalent (chromous) ion is rapidly oxidized to the trivalent (Cr(III); chromic] form." [Nordberg, p. 718] "Bivalent compounds are unstable and have little commercial value." [Asthma in the Workplace, p. 296] May be harmful after ingestion, inhalation, or skin contact; May damage eyes; "Stable under recommended storage conditions." [MSDSonline] A skin and respiratory tract irritant; [ICSC] See "Chromium."
Biomedical References
Exposure Assessment
PEL (OSHA)
0.5 mg/m3, as Cr
Explanatory Notes
VP = practically zero; Mp = 1900 deg C; [MSDSonline]
Diseases, Processes, and Activities Linked to This Agent
Processes
Industrial Processes with risk of exposure: