Dialifor
Agent Name
Dialifor
CAS Number
10311-84-9
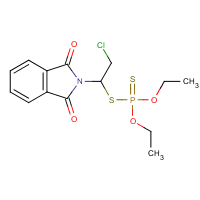
Formula
C14-H17-Cl-N-O4-P-S2
Major Category
Pesticides
Synonyms
Dialifos; Dialiphor; Dialiphos [ISO-French]; ENT 27320; Hercules 14503; N-(2-Chloro-1-(diethoxyphosphinothioylthio)ethyl)phthalimide; O,O-Diaethyl-S-2-chlor-1-(phthalimido)-aethyl-dithiophosphat [German]; O,O-Diethyl S-(2-chloro-1-phthalimidoethyl)phosphorodithioate; O,O-Diethyl phosphorodithioate S-ester with N-(2-chloro-1-mercaptoethyl) phthalimide; O,O-Diethyl phosphorodithioate S-ester with N-(2-chloro-1-mercaptoethyl)phthalimide; Phosphorodithioic acid, O,O-diethyl ester, S-ester with N-(2-chloro-1-mercaptoethyl)phthalimide; Phosphorodithioic acid, S-(2-chloro-1-(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)ethyl) O,O-diethyl ester; Phosphorodithioic acid, S-(2-chloro-1-phthalimidoethyl) O,O-diethyl ester; Phosphorodithioic acid, S-(2-chloro-1-phthalimidoethyl)-O,O-diethyl ester; S-(2-Chloro-1-(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)ethyl) O,O-diethyl phosphorodithioate (9CI); S-(2-Chloro-1-(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)ethyl)O,O-diethyl phosphorodithioate; S-(2-Chloro-1-phthalimidoethyl) O,O-diethyl phosphorodithioate; S-(2-Chloro-1-phthalmidoethyl)-O,O-diethylphosphorothionate; Torak; [ChemIDplus]
Category
Organophosphate Insecticides
Description
White solid; May also be liquid; [HSDB] Colorless solid; [MSDSonline] Pure: White solid; Isolated technical: Brown solid; Commercial technical: Brown liquid; [INCHEM JMPR]
Sources/Uses
Used as acaricide and insecticide for fruits, nuts, potatoes, vegetables, cotton, and other crops; [HSDB]
Comments
A cholinesterase inhibitor; [Hawley] A mild skin and eye irritant; In an exposure of 120 vineyard workers, symptoms included dizziness, weakness, headache, blurred vision, and tightness in the chest; “Most plasma and red cell values were depressed more than 60%.” All fully recovered, although 85 received medical attention and three were hospitalized; [HSDB] Some evidence of reproductive effects in feeding study with hamsters; [RTECS] No adverse gross or histopathological observations in three generation oral study with rats; No teratogenic effects observed in two studies with rabbits and one with macaques; No evidence of mutagenicity; No clinical observations of neurotoxicity in study with birds; Mildly irritating to skin and eyes of rabbits; Effects in high-dose feeding studies with rodents include cholinesterase inhibition and some evidence of liver necrosis; No significant effects observed in gross, histopathological, or tumor frequency in long term feeding studies with rats; [INCHEM JMPR] “The average of two baseline respective cholinesterase activity determinations three days apart, with no exposures to enzyme inhibiting pesticides for at least 30 days, is recommended for each worker prior to exposure to cholinesterase inhibitors because of large inter-individual differences in published baseline values. To be established at least once a year. Removal from workplace exposures is recommended until the cholinesterase activity returns to within 20% of baseline.” [TLVs and BEIs]
Restricted
Cancelled--no longer registered as a pesticide for use in the US; [HSDB]
Biomedical References
Exposure Assessment
BEI
Acetylcholinesterase activity in red blood cells = 70% of individual's baseline; Butylcholinesterase activity in serum or plasma = 60% of individual's baseline; Sample at end of shift; [TLVs and BEIs]
Vapor Pressure
6.2E-08 mm Hg
Adverse Effects
Other Poison
Organophosphate
Diseases, Processes, and Activities Linked to This Agent
Diseases
Occupational diseases associated with exposure to this agent:
Processes
Industrial Processes with risk of exposure: