Sodium chromate
Agent Name
Sodium chromate
CAS Number
7775-11-3
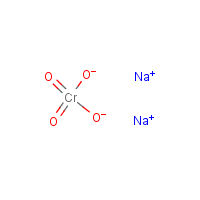
Formula
Cr-H2-O4.2Na
Major Category
Metals
Synonyms
Chromate of soda; Chromic acid (H2(sup 51)CrO4), disodium salt; Chromic acid (H2CrO4), disodium salt; Chromium disodium oxide; Chromium sodium oxide (CrNa2O4); Disodium chromate; Disodium chromate (Na2(sup 51)CrO4); Neutral sodium chromate; Rachromate; Sodium chromate(VI); Chromic acid, disodium salt; [ChemIDplus] UN3288
Category
Chromium Compounds, Inorganic
Description
Yellow hygroscopic solid; Soluble in water; [ICSC] Yellow crystals; [MSDSonline] Hygroscopic--forms several hydrates; [Ullmann]
Sources/Uses
Used to make pigments, inks, coatings, and other chromates; Also used in metal treatment, dyeing (mordant), leather tanning, corrosion inhibition, chlorate manufacturing, and aluminum etching; Use as fungicide (wood preservative) cancelled in the US by the EPA; [HSDB] Used in the textile (dyeing auxiliary) and petroleum (corrosion inhibitor) industries; [Ullmann]
Comments
A corrosive substance that can cause injury to the skin, eyes, and respiratory tract; Can be absorbed through skin; May cause skin sensitization and asthma after prolonged contact; May cause kidney and liver injury; May cause perforated nasal septum; [ICSC] Can cause burns; Very toxic by inhalation; May cause skin and respiratory sensitization after prolonged contact; [eChemPortal: ESIS] See "Chromium" and linked occupational diseases.
Biomedical References
Exposure Assessment
Skin Designation (ACGIH)
Yes
TLV (ACGIH)
0.0001 mg/m3, as Cr(VI), inhalable particulate matter
STEL (ACGIH)
0.0005 mg/m3, as Cr(VI), inhalable particulate matter
PEL (OSHA)
0.005 mg/m3, as Cr(VI)
Lethal Concentration
LC50 (rat) = 33 mg/m3/4h
Explanatory Notes
The Guide in the Emergency Response Guidebook is for "Toxic solid, inorganic, n.o.s."
Adverse Effects
Skin Sensitizer
Yes
Asthma
Yes
Hepatotoxin
Hepatoxic (a) from occupational exposure (secondary effect) or (b) in animal studies or in humans after ingestion
Nephrotoxin
Yes
Dermatotoxin
Skin burns
IARC Carcinogen
Established
ACGIH Carcinogen
Confirmed Human
Diseases, Processes, and Activities Linked to This Agent
Diseases
Occupational diseases associated with exposure to this agent:
Processes
Industrial Processes with risk of exposure: