Ammonium bisulfide
Agent Name
Ammonium bisulfide
CAS Number
12124-99-1
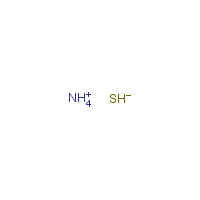
Formula
H4-N.H-S
Major Category
Other Classes
Synonyms
Ammonium hydrogen sulfide; Ammonium hydrosulfide; Ammonium mercaptan; Ammonium sulfhydrate; Ammonium sulfide; Monoammonium sulfide; Sirnik amonny [Czech]; Ammonium hydrogensulphide; [ChemIDplus] UN2683
Category
Sulfur Compounds
Description
White to yellow hygroscopic solid; [ICSC] Decomposes when moisture or >0 deg C; [Hawley] Technical grade is 40-44% and kept basic with sodium hydroxide; [CAMEO] Solution has strong odor of rotten eggs and ammonia; [CHRIS]
Sources/Uses
Used in lubricants; [Merck Index] Used in photography, textiles, synthetic flavors, for coloring brasses and bronzes, and for iron control (soda ash production); [CAMEO]
Comments
Crystals decompose at room temperature forming hydrogen sulfide (may be fatal) and ammonia; A severe skin irritant; [Merck Index] A severe skin, eye, and respiratory tract irritant; Can be absorbed through skin; [ICSC] Produces hydrogen sulfide on mixing with water; A reducing agent; [CAMEO] Aqueous solution: Causes severe eye burns and severe skin irritation; Inhalation of 500 ppm for 30 minutes causes headache, dizziness, and bronchial pneumonia; 600 ppm for 30 minutes can be fatal; Can be absorbed through skin resulting in hydrogen sulfide poisoning; [CHRIS] Causes mydriasis, dyspnea, and respiratory stimulation in lethal-dose animal studies (dermal, intraperitoneal, intravenous, and oral routes); [RTECS] See "Hydrogen sulfide."
Biomedical References
Exposure Assessment
Vapor Pressure
390 mm Hg
Explanatory Notes
The Guide in the Emergency Response Guidebook is for "Ammonium sulfide, solution."
Adverse Effects
Dermatotoxin
Skin burns
Other Poison
Chemical Asphyxiant
Diseases, Processes, and Activities Linked to This Agent
Diseases
Occupational diseases associated with exposure to this agent:
Processes
Industrial Processes with risk of exposure:
Activities
Activities with risk of exposure: