Ethylene glycol
Agent Name
Ethylene glycol
CAS Number
107-21-1
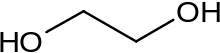
Formula
C2-H6-O2
Major Category
Other Classes
Synonyms
1,2-Dihydroxyethane; 1,2-Ethanediol; 146AR; 2-Hydroxyethanol; Aethylenglykol [German]; Dowtherm SR 1; Ethylene alcohol; Ethylene dihydrate; Ethylene glycol; Fridex; Glycol; Glycol alcohol; Glycol, ethylene-; Lutrol-9; Macrogol 400 BPC; Monoethylene glycol; Norkool; Ramp; Tescol; Ucar 17; Union Carbide XL 54 Type I De-icing Fluid; Zerex; [ChemIDplus] UN8027
Category
Ethylene Glycols
Description
Clear, colorless, syrupy, odorless liquid. [antifreeze] [Note: A solid below 9 degrees F.]; [NIOSH]
Sources/Uses
Used in antifreeze and deicing solutions for cars, boats, and aircraft; also used as a solvent for paints, plastics, photographic developing solutions, coolants, hydraulic fluids, and inks; [ATSDR ToxFAQs] Highest risk for exposure in deicing aircraft and runways; [Reference #2] Occupational exposure mainly through skin and eyes, but also inhalation if heated or aerosolized; 50% solution used to de-ice bridges; Has been used in adhesives, polishes, cosmetics, and pharmaceuticals; [ACGIH]
Comments
TLV Basis is upper respiratory tract irritation; The oral lethal dose in adult humans is about 1.4 ml/kg or about 100 ml. No adverse effects to human eyes after 4 weeks of exposure to 6.6 ppm; [ACGIH] Ethylene glycol is a skin irritant and a poison by ingestion. No health effects have been reported in persons chronically exposed to levels of ethylene glycol found in the environment. [ATSDR Case Studies #30] "Toxic inhalation of ethylene glycol is unlikely at room temperature because of the chemical's low volatility, but can occur when the liquid is heated, agitated, or sprayed." Toxicity after ingestion includes inebriation, metabolic acidosis, and renal failure. [ATSDR Medical Management] Ethylene glycol is not significantly absorbed through the skin. Poisoning after inhalation exposure is "unlikely." [Ford, p. 758] Combined osmolar and anion gaps suggest poisoning by methanol or ethylene glycol, but also may occur in severe alcoholic ketoacidosis or diabetic ketoacidosis. [Olson, p. 35] A skin, eye, and respiratory tract irritant; May cause effects on the kidneys and CNS; [ICSC] Causes acute tubular necrosis after ingestion but not after occupational exposures because of low vapor pressure; [Rosenstock, p. 1006]
Biomedical References
Exposure Assessment
Skin Designation (ACGIH)
No
TLV (ACGIH)
25 ppm, vapor fraction
STEL (ACGIH)
50 ppm, vapor fraction (10 mg/m3, inhalable particulate matter, aerosol only
MAK
10 ppm
Vapor Pressure
0.05 mm Hg
Odor Threshold Low
159 ppm
Lethal Concentration
LC (rat) > 200 mg/m3/4h
Explanatory Notes
Flash point = 232 deg F; Odor threshold; [ACGIH] VP from HSDB;
Half Life
Serum: 2-3 hours; not detectable in urine or tissues after 24-48 hours (metabolites present for longer periods); [TDR, p. 659]
NFPA
must be preheated
ERPG-1
50 ppm
ERPG-2
200 ppm
ERPG-3
300 ppm
Adverse Effects
Neurotoxin
Other CNS neurotoxin
Nephrotoxin
Yes
ACGIH Carcinogen
Not Classifiable
Diseases, Processes, and Activities Linked to This Agent
Diseases
Occupational diseases associated with exposure to this agent:
Processes
Industrial Processes with risk of exposure: