Sodium hypochlorite
Agent Name
Sodium hypochlorite
CAS Number
7681-52-9
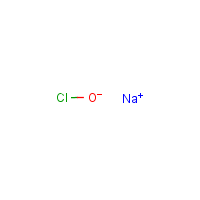
Formula
Cl-O.Na
Major Category
Other Uses
Synonyms
AD Gel; Antiformin; B-K liquid; Carrel-Dakin solution; Chloros; Chlorox; Cloralex; Cloropool; Clorox; Clorox liquid bleach; Dakin's solution; Dakins half; Dakins quarter; Dakins solution; Deosan; Deosan Green Label Steriliser; Di-Dak-Sol; Dispatch; Hospital Milton; Household bleach; Hyclorite; Hypochlorite sodium; Hyposan and Voxsan; Hypure; Hypure N; Javel water; Javelle water; Javex; Klorocin; Milton; Milton Crystals; Modified Dakin's solution; Neo-cleaner; Neoseptal CL; Parozone; Purin B; Sodium hypochlorite [solution, diluted]; Sodium oxychloride; Solutions, Dakin's; Sunnysol 150; Surchlor; XY 12; Youxiaolin; Chlorinated water (sodium hypochlorite); Hypochlorous acid, sodium salt; Sodium hypochlorite solution; Sodium hypochlorite solution (15% or less); [ChemIDplus] UN1791
Category
Biocides/Disinfectants
Description
Greenish-yellow liquid with a disagreeable, sweetish odor; [HSDB] Produced as the pentahydrate; Solid with a mp of 18 deg C; [Merck Index]
Sources/Uses
Used as a disinfectant and bleaching agent with applications in the chemical, paper, textile, water treatment, and dairy industries; it is used to disinfectant water in swimming pools and cooling towers of power plants. [HSDB] 5% solution used as a hypo eliminator in photo development; [www.ci.tucson.az.us/arthazards/medium.html] Used in the EU on mushroom crops; Also used in the paint, lime, glass, pharmaceutical, metal (detoxify cyanide baths), and synthetics industries; [EFSA]
Comments
Corrosive to skin; [Quick CPC] Addition of acid to hypochlorite solution releases chlorine gas; Addition of ammonia releases chloramine gas; High inhalation exposure, especially if in confined space, can cause pulmonary edema; Ingestion can cause methemoglobinemia; Ingestion or 3% to 5% solution will cause burning of mouth and throat without other injury expected; [HSDB] Household bleaches (approximately 5% sodium hypochlorite) are irritants; more concentrated bleaches (10-15% sodium hypochlorite) are corrosive. [ICSC] The pentahydrate is decomposed by carbon dioxide in the air; The anhydrous form is highly explosive; [Merck Index] Only commercially produced as an aqueous solution; [EFSA] See "Hypochlorous acid."
Reference Link #1
Biomedical References
Exposure Assessment
Explanatory Notes
The estimated odor threshold is the same as chlorine, 0.3 ppm; [AIHA] The Guide in the Emergency Response Guidebook is for "Hypochlorite solution."
Reference Link #2
WEEL
STEL = 2 mg/m3
Adverse Effects
Toxic Pneumonitis
Yes
Methemoglobinemia
MetHgb is secondary toxic effect
Dermatotoxin
Skin burns
Diseases, Processes, and Activities Linked to This Agent
Processes
Industrial Processes with risk of exposure:
- Electroplating
- Farming (Respiratory Hazards)
- Glass Manufacturing
- Metal Extraction and Refining
- Petroleum Production and Refining
- Photographic Processing
- Pulp and Paper Processing
- Sewer and Wastewater Treatment
- Textiles (Fiber & Fabric Manufacturing)
- Textiles (Printing, Dyeing, or Finishing)
- Using Disinfectants or Biocides
Activities
Activities with risk of exposure: