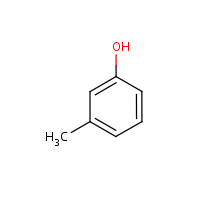
m-Cresol
Agent Name
m-Cresol
Alternative Name
3-Methylphenol
CAS Number
108-39-4
Formula
C7-H8-O
Major Category
Other Classes
Synonyms
1-Hydroxy-3-methylbenzene; 1-Methyl-3-hydroxybenzene; 3-Cresol; 3-Hydroxytoluene; 3-Methylphenol; Bacticin; Celcure Dry Mix (chemicals for wood preserving); Franklin Cresolis; Metacresol; Rover's Dog Shampoo; m-Cresylic acid; m-Hydroxytoluene; m-Kresol [German]; m-Methylphenol; m-Oxytoluene; m-Toluol; meta-Cresol; Cresol, m-; Cresol, m-isomer; Phenol, 3-methyl-; [ChemIDplus] m-Cresole; m-Cresylic; meta-Cresylic acid; Hydroxy-3-methylbenzene; 3-Methyl-1-hydroxybenzene; m-Methylphenylol; [HSDB] UN2076
Category
Phenols
Description
Colorless or yellowish liquid with a phenolic odor; mp = 11-12 deg C; [Merck Index] Hygroscopic; [CAMEO]
Sources/Uses
Used to make synthetic resins; Also used in disinfectants, photographic developers, explosives, fungicides, and as an industrial solvent; [Merck Index] Used in combination with 2,4-xylenol (CAS# 105-67-9) as a bactericide/bacteriostat for the control of crown gall, olive knot, and burr knot in fruit, nut, and ornamental trees and vines; [EPA REDs] Used as flavoring agent or adjuvant; [FDA] Used in ore flotation, fumigation compounds, degreasing compounds, paintbrush cleaners, lubricating oils, as a wood preservative, engine and metal cleaner, and in the textile industry; [NTP] Naturally occurring in many plants, petroleum, and, coal tar, as well as emissions from the combustion of wastes, coal, wood, vehicles, and cigarettes; Used primarily as an intermediate for synthetic vitamin E, pesticides, fragrances, antioxidants, disinfectants, and preservatives; Used as a pharmaceutical bactericide and preservative; Uses for all cresol isomers include as preservatives or stabilizers in cleaning/washing agents, surface treatment products, paints, adhesives, binding agents, corrosion inhibitors, and impregnation materials; [OECD SIDS: m-/p-Cresol Category - 2003]
Comments
Causes burns; Inhalation may cause pulmonary edema; Exposure may cause CNS depression and hemolytic anemia; Can be absorbed through skin; [ICSC] May cause second degree burns after a few minutes contact; May cause liver and kidney injury; [CHRIS] Least toxic of the three cresol isomers; May cause skin sensitization; [HSDB] No evidence of skin sensitization in limited studies of humans and guinea pigs using p-cresol and m-/p-cresol; A survey reports hypersensitivity reactions of certain individuals to cresol (unspecified isomer); Neither isomer produced mortality or clinical signs of toxicity following exposure to saturated vapor concentrations, although inhalation of aerosols may be fatal; [OECD SIDS: m-/p-Cresol Category - 2003] Safe when used as a flavoring agent in food; [JECFA] Causes burns; Inhalation may cause corrosive injuries to upper respiratory tract and lungs; Toxic by ingestion and skin absorption; Targets the CNS, lungs, liver, kidney, and eyes; [Sigma-Aldrich MSDS] See "Cresol, all isomers."
Reference Link #1
Biomedical References
Exposure Assessment
Skin Designation (ACGIH)
Yes
TLV (ACGIH)
20 mg/m3, inhalable fraction and vapor
PEL (OSHA)
22 mg/m3
Vapor Pressure
0.11 mm Hg
Odor Threshold Low
5 ppm
Lethal Concentration
LC50 (rat) > 710 mg/m3/1hr
Explanatory Notes
Odor threshold from NTP; The Guide in the Emergency Response Guidebook is for "Cresols."
NFPA
high ambient temp required
Adverse Effects
Toxic Pneumonitis
Yes
Hepatotoxin
Hepatoxic (a) from occupational exposure (secondary effect) or (b) in animal studies or in humans after ingestion
Dermatotoxin
Skin burns
ACGIH Carcinogen
Not Classifiable
Diseases, Processes, and Activities Linked to This Agent
Processes