Sodium carbonate
Agent Name
Sodium carbonate
CAS Number
497-19-8
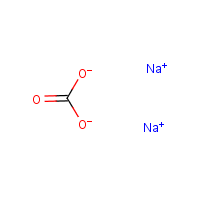
Formula
C-H2-O3.2Na
Major Category
Metals
Synonyms
Carbonic acid, disodium salt; Soda Ash; Carbonic acid disodium salt; Bisodium carbonate; Calcined soda; Carbonic acid sodium salt; Carbonic acid, disodium salt; Crystol carbonate; Disodium carbonate; Dynamar L 13890; Light Ash; Na-X; Natrium Carbonicum Calcinatum; Natrium Carbonicum Siccatum; Snowlite I; Soda ash; Soda Ash Light 4P; Soda, calcined; Sodium carbonate, anhydrous; Sodium Carbonate, Anhydrous ASTM D458; Sodium Carbonate, Anhydrous GE Materials D4D5; Solvay soda; Suprapur 6395; Trona; V 20N; V Soda; Washing soda; [ChemIDplus] Sodium carbonate (anhydrous); [ICSC]
Category
Metals, Inorganic Compounds
Description
White or grayish-white odorless solid; Hygroscopic; [HSDB] White hygroscopic powder; [Sigma-Aldrich MSDS]
Sources/Uses
Used to make glass and inorganic sodium compounds, in detergents and cleansers, pulp and paper production, water treatment, aluminum production, textile processing (cleaning, bleaching, and dye assist formulation), petroleum refining, coal liquefaction, photography, mining (uranium and lithium recovery), flue gas desulfurization, decontaminating radioactive surfaces, sealing ponds from leakage, effluent and acid waste neutralization, consumer products (i.e. cosmetics, scouring powders, and clothes, dish, and floor cleaners), boiler water compounds, as food additive, analytical reagent, pharmaceutic aid (alkalizing agent), and in human and veterinary medicine; [HSDB] Used as a fungicide in institutional, commercial, and household surface disinfectants/sanitizers; Also used as an inert ingredient in over 600 active pesticides in the US; Generally regarded as safe (GRAS) by the FDA; [Reference #1] Used as a flavoring agent, antioxidant, pH control agent, curing or pickling agent, and processing aid for foods; [FDA] Used in hide preparation for leather production; [PMID 21938525]
Comments
Occurs naturally as the hydrate (thermonatrite) and the decahydrate (natron or natrite); Technical material referred to as soda ash (approximately 99% pure); A skin irritant; Overexposure to dust or vapor causes mucous membrane irritation, coughing, and shortness of breath (chronic overexposure may cause nasal septum perforation); The monohydrate (CAS# 5968-11-6) and decahydrate (CAS# 6132-02-1) are also commercial products; [Merck Index] Skin irritation is none to mild; Dust or concentrated solutions are moderate to severe eye irritants; Cases of nasal septal perforation have been reported in men with continuous exposure to soda ash dust. [CHEMINFO] An alkaline corrosive agent used as detergent in dishwashers; [Olson, p. 214] A skin, eye, and respiratory tract irritant; Prolonged or repeated exposure may cause perforation of the nasal septum; [ICSC] Acceptable daily intake (ADI) as food additive not limited; [JECFA] A mild skin and severe eye irritant; [Sigma-Aldrich MSDS] See "Sodium carbonate decahydrate" and "Sodium carbonate monohydrate."
Reference Link #1
Biomedical References
Exposure Assessment
Lethal Concentration
LC50 (rat) = 2,300 mg/m3/2hr
Adverse Effects
Dermatotoxin
Skin burns
Diseases, Processes, and Activities Linked to This Agent
Processes
Industrial Processes with risk of exposure:
- Aluminum Producing
- Glass Manufacturing
- Leather Tanning and Processing
- Mining
- Petroleum Production and Refining
- Photographic Processing
- Pulp and Paper Processing
- Sewer and Wastewater Treatment
- Textiles (Fiber & Fabric Manufacturing)
- Textiles (Printing, Dyeing, or Finishing)
- Using Disinfectants or Biocides
Activities
Activities with risk of exposure: